2018
Journal Publication
Developmental Biology
Cellular, ultrastructural and molecular analyses of epidermal cell development in the planarian Schmidtea mediterranea.
Cheng LC, Tu KC, Seidel CW, Robb SMC, Guo F, Sánchez Alvarado A
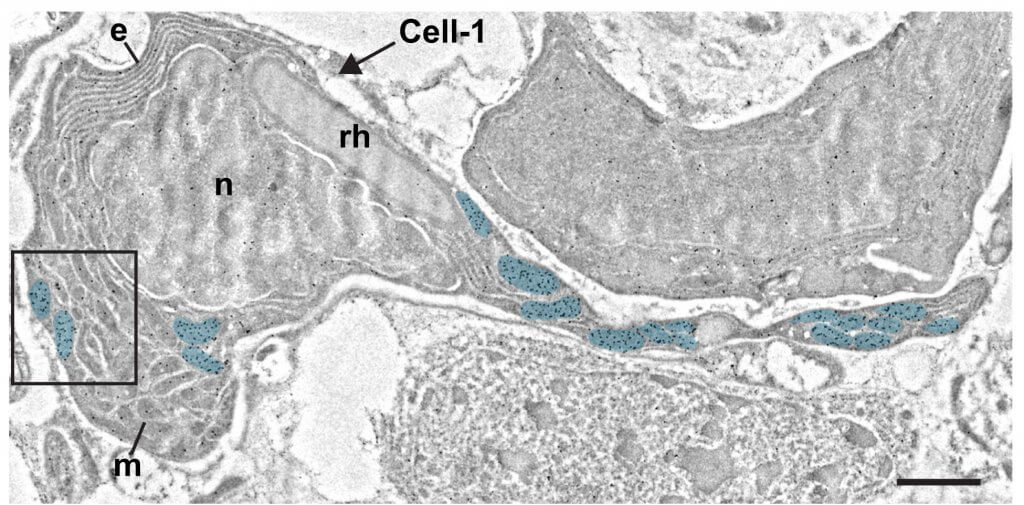
The epidermis is essential for animal survival, providing both a protective barrier and cellular sensor to external environments. The generally conserved embryonic origin of the epidermis, but the broad morphological and functional diversity of this organ across animals is puzzling. We define the transcriptional regulators underlying epidermal lineage differentiation in the planarian Schmidtea mediterranea, an invertebrate organism that, unlike fruitflies and nematodes, continuously replaces its epidermal cells. We find that Smed-p53, Sox and Pax transcription factors are essential regulators of epidermal homeostasis, and act cooperatively to regulate genes associated with early epidermal precursor cell differentiation, including a tandemly arrayed novel gene family (prog) of secreted proteins. Additionally, we report on the discovery of distinct and previously undescribed secreted organelles whose production is dependent on the transcriptional activity of soxP-3, and which we term Hyman vesicles.
Address reprint requests to: Alejandro Sánchez Alvarado